Charkowski, A. O. 2018. The changing face of bacterial soft-rot dis-eases.
Annu. Rev. Phytopathol. 56: 269-288.


Food and Agriculture Organization Corporate Statistical Database. 2021. Export news. URLhttps://www.fao.org/faostat/en/#data/QCL. [11 July 2023].
Gottsberger, R. A. and Huss, H. 2016.
Pectobacterium carotovorum subsp.
brasiliensis causing a soft rot on styrian oil pumpkin in Austria.
New Dis. Rep. 33: 12.

Hua, G. K. H., Ali, E. and Ji, P. 2020. Characterization of bacterial pathogens causing fruit soft rot and stem blight of bell pepper in Georgia, USA.
J. Plant Pathol. 102: 311-318.

Hyman, L. J., Sullivan, L., Toth, I. K. and P├®rombelon, M. C. M. 2001. Modified crystal violet pectate medium (CVP) based on a new polypectate source (Slendid) for the detection and isolation of soft rot erwinias.
Potato Res. 44: 265-270.

Jaramillo, A., Huertas, C. A. and G├│mez, E. D. 2017. First report of bacterial stem rot of tomatoes caused by
Pectobacterium carotovorum subsp.
brasiliense in Colombia.
Plant Dis. 101: 830.

Kim, Y. J., Hwang, A. J., Noh, J. J., Wang, X., Lee, J. E., Yoo, E. et al. 2021. ╬▓-carotene, cucumisin content and fruit morphology of melon (Cucumis melo L.) germplasm collections. Korean J. Plant Res. 34: 555-565.
Meng, X., Chai, A., Shi, Y., Xie, X., Ma, Z. and Li, B. 2017. Emergence of bacterial soft rot in cucumber caused by
Pectobacterium carotovorum subsp.
brasiliense in China.
Plant Dis. 101: 279-287.


Naas, H., Sebaihia, M., Orfei, B., Rezzonico, F., Buonaurio, R. and Moretti, C. 2018.
Pectobacterium carotovorum subsp.
brasiliense and
Pectobacterium carotovorum subsp.
carotovorum as causal agents of potato soft rot in Algeria.
Eur. J. Plant Pathol. 151: 1027-1034.

Onkendi, E. M. and Moleleki, L. N. 2014. Characterization of
Pectobacterium carotovorum subsp.
carotovorum and
brasiliense from diseased potatoes in Kenya.
Eur. J. Plant Pathol. 139: 557-566.

Panda, P., Fiers, M. A. W. J., Armstrong, K. and Pitman, A. R. 2012. First report of blackleg and soft rot of potato caused by
Pectobacterium carotovorum subsp.
brasiliensis in New Zealand.
New. Dis. Rep. 26: 15.

Park, K. T., Hong, S. M., Back, C. G., Kang, I. K., Lee, S. Y., Ten, L. N. et al. 2023.
Pectobacterium brasiliense as a causative agent for soft rot of radish in Korea.
Res. Plant Dis. 29: 64-71.

Park, K. T., Hong, S. M., Back, C. G., Kim, S. Y., Lee, S. Y., Kang, I. K. et al. 2022. First report of
Pectobacterium brasiliense causing soft rot on graft cactus in Korea.
Res. Plant Dis. 28: 172-178.

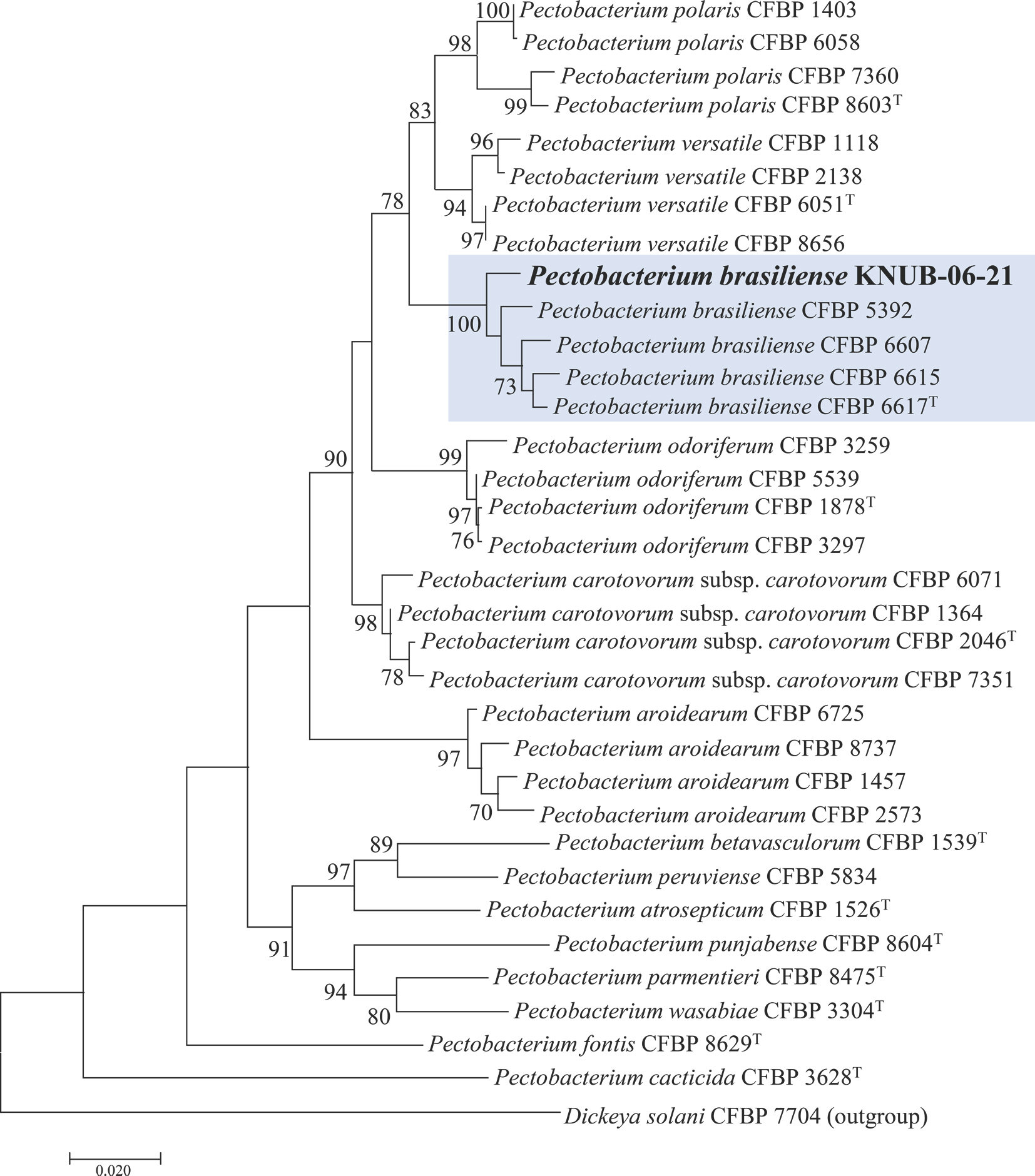
Portier, P., P├®dron, J., Taghouti, G., Dutrieux, C. and Barny, M. A. 2020. Updated taxonomy of
Pectobacterium genus in the CIRM-CFBP bacterial collection: when newly described species reveal ŌĆ£OldŌĆØ endemic population.
Microorganisms 8: 1441.



Portier, P., P├®dron, J., Taghouti, G., Fischer-Le Saux, M., Caullireau, E., Bertrand, C. et al. 2019. Elevation of
Pectobacterium carotovorum subsp.
odoriferum to species level as
Pectobacterium odoriferum sp. nov., proposal of
Pectobacterium brasiliense sp. nov. and
Pectobacterium actinidiae sp. nov., emended description of
Pectobacterium carotovorum and description of
Pectobacterium versatile sp. nov., isolated from streams and symptoms on diverse plants.
Int. J. Syst. Evol. Microbiol. 69: 3207-3216.


Yi, Y. K. and Kim, R. H. 1996. Occurrence of bacterial soft rot of melon caused by Erwinia carotovora subsp. carotovora. Korean J. Plant Pathol. 12: 116-120.
Zlatkovi─ć, N., Proki─ć, A., Ga┼Īi─ć, K., Kuzmanovi─ć, N., Ivanovi─ć, M. and Obradovi─ć, A. 2019. First report of
Pectobacterium carotovorum subsp.
brasiliense causing soft rot on squash and watermelon in Serbia.
Plant Dis. 103: 2667.







 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Supplement
Supplement Print
Print